iEXCEL Implant System – The ultimate dental performance system
NEW CUSTOMER OFFER ONLY - Provide your best contact information and let us handle the work while you enjoy the benefits!
*General Terms and Conditions of Purchase apply. No substitution, no partial returns.
Additional taxes, training and shipping charges may apply. Offer cannot be combined with any other discounts or promotions. Offer valid through December 31, 2026.
Straumann collects your information to facilitate the commercial relationship, contact you regarding future product offerings and promotion and all other permissible business purposes. Please see our Privacy Policy for more information.
WHY iEXCEL?
Harness streamlined workflows and advanced technologies to help support achievement of immediate, predictable outcomes.
A TorcFit™ connection that facilitates the transfer of high torques for a precise implant abutment fit
Dynamic Bone Management helps make immediate protocols achievable and predictable, even in tough cases.
New slim-apex tapered implants support less-invasive treatment* and are designed for efficient, stable insertion.
*If a guided bone regeneration procedure can be avoided.
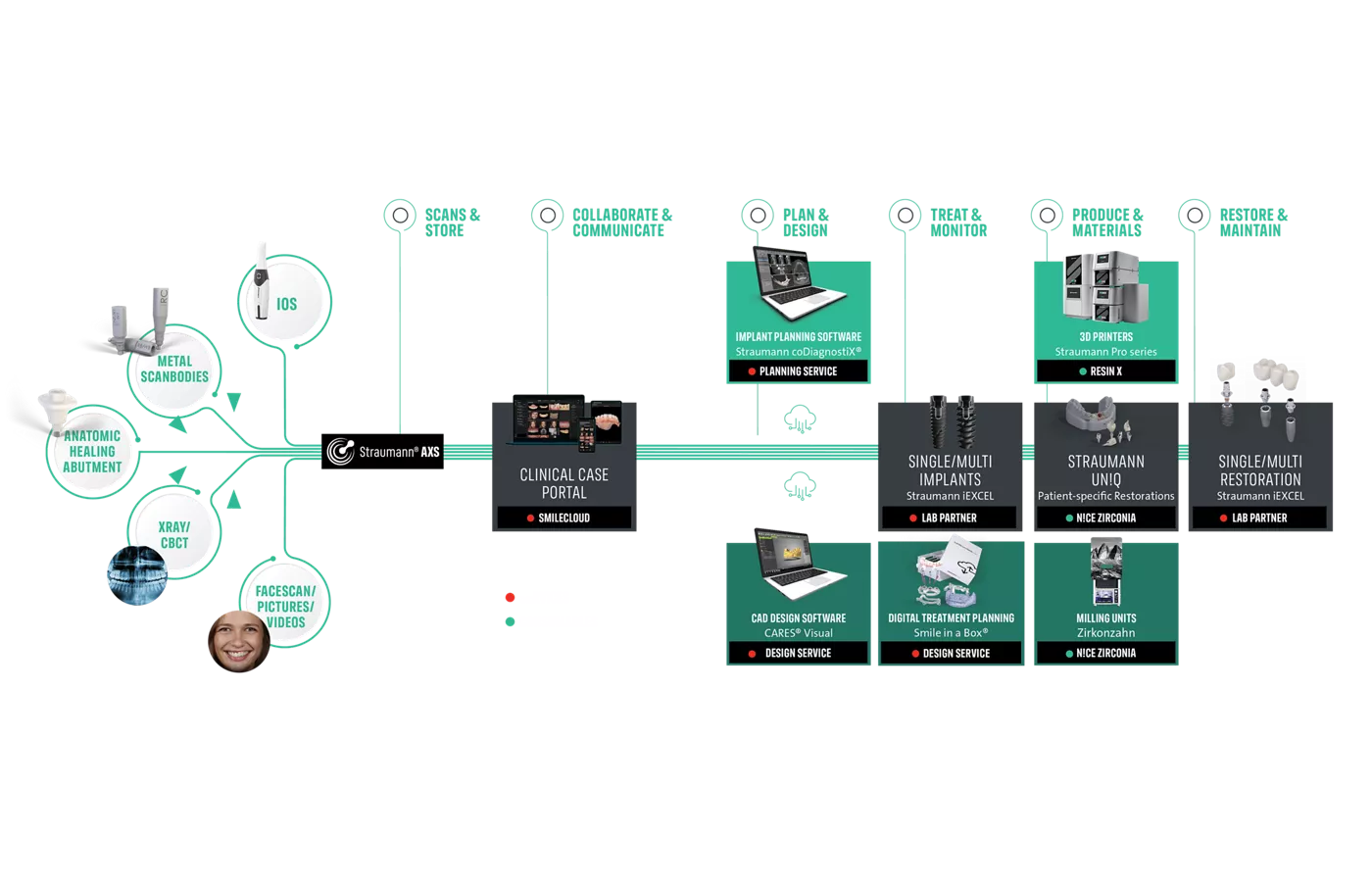
END-TO-END INTERDISCIPLINARY DIGITAL TREATMENT WORKFLOW
Experience the evolution of treatment planning and execution with our end-to-end Interdisciplinary digital treatment workflow. This comprehensive approach supports continuity of care across disciplines. From imaging to treatment delivery, target new levels of precision, efficiency, and patient satisfaction.
Hear directly from some of your colleagues about their experiences today—so you don’t just have to take our word for it!
For accurate product usage, please refer to the Instructions for Use (IFU) at ifu.straumann.com. Straumann is not responsible for any improper use of implants or products based on customer case repost.