
What is this about?
- A modern treatment approach for peri-implantitis is explained and how it performs in real clinical practice.
- It focuses on electrolytic cleaning (GalvoSurge®) as an innovative method for implant surface decontamination.
- Scientific evidence and real-world data to evaluate clinical effectiveness and outcomes is summarized.
Growing insights, evolving disease definitions, and innovative treatment approaches have expanded therapeutic possibilities for peri-implantitis, a condition of increasing relevance in contemporary implant dentistry. At the same time, an expanding body of evidence continues to clarify factors influencing disease onset, progression, prevention, and treatment.
Among recent developments, a novel treatment concept - electrolytic cleaning - has emerged for the decontamination of titanium implants affected by peri-implantitis1–3. This approach is based on the application of physiologically harmless, low-voltage currents, combined with continuous irrigation of the implant surface with a buffered electrolyte solution 4–7. The applied current induces a controlled electrochemical process along the entire implant surface, independent of accessibility or implant macro- and microdesign6, and the resulting in-situ formation of small amounts of hydrogen facilitates the detachment of biofilm7-8. While electrolytic principles have been used in other fields for in-situ disinfectant generation9, their standardized application in peri-implantitis treatment has only recently been established under the name GalvoSurge®.
The efficacy of electrolytic cleaning has been demonstrated in both preclinical and clinical studies. In vitro investigations showed superior decontamination of multispecies biofilms compared to conventional methods, achieving near-sterile implant surfaces and preventing subsequent bacterial growth, while not adversely affecting surface nor the corrosion resistance of the treated implants6,8,11-13. In a preclinical ligature-induced peri-implantitis model, electrolytic cleaning resulted in more favorable healing outcomes than conventional approaches, indicating the potential for complete re-osseointegration4. Clinical studies reported consistent resolution of inflammation and peri-implantitis, substantial radiographic bone regeneration and defect fill, and significant improvements in clinical parameters such as probing depth, bleeding on probing, and suppuration, with outcomes influenced by defect morphology and sustained over time 5, 9,14,15. These findings are further supported by human histological analyses and studies of explanted implants, confirming that electrolytically treated surfaces can promote conditions conducive to complete re-osseointegration9, 10,16,17.
Real-world data are essential for understanding how treatments perform in everyday clinical practice, beyond the controlled conditions of clinical trials. They capture the variability in patient populations, clinical decision-making, and treatment complexity, providing a more comprehensive picture of clinical effectiveness and safety. Even though real-world data have inherent limitations, such as the lack of randomization and controlled comparisons, they play a key role in informing clinical practice, supporting evidence-based decisions, and complementing findings from traditional clinical studies. In this context, Straumann conducted a multicenter, real-world data collection survey based on retrospective evaluation of patient charts to assess the performance of treatments involving GalvoSurge® as part of peri-implantitis management under routine clinical conditions18.
The survey was conducted between April and December 2025 using a structured online questionnaire, supported by standardized templates to ensure consistency in data reporting. It included eight centers across five European countries, representing both private practices and academic clinical settings:
- Switzerland: Dr. Daniel Wolf (Luzern)
- Lithuania: Dr. Algirdas Puišys (Vilnius)
- Belgium: Dr. Bruno De Carvalho (University of Liège)
- Spain: Dr. Alberto Monje (Badajoz) and Dr. Ramón Pons (Barcelona)
- Italy: Prof. Massimo Frosecchi (Florence), Dr. Mario Roccuzzo (Turin), and Dr. Nicola de Angelis (Acqui Terme)
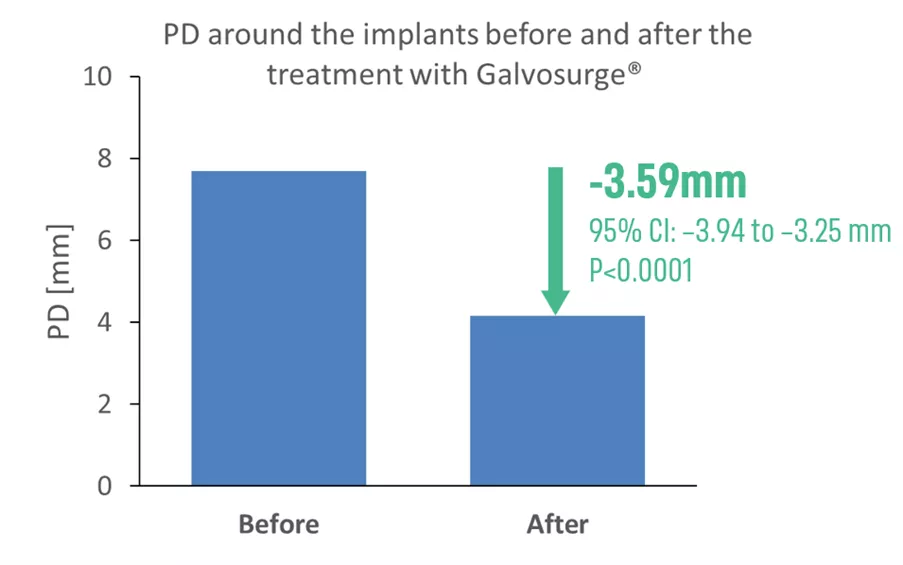
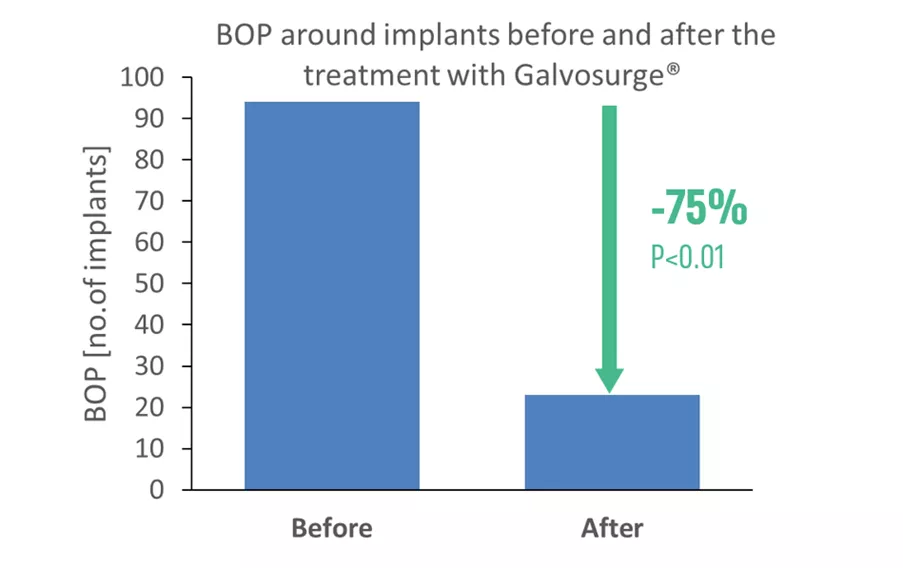
A total of 89 patients and 104 implants were included in the analysis. The study population included both male and female patients, with the majority aged 40 years and older. The treated implants mainly represented advanced peri-implantitis cases, with a mean baseline probing depth (PD) of 7.69 mm and 94 implants displaying bleeding on probing (BOP). Implants were distributed across both the maxilla and mandible, in anterior and posterior regions, and supported single as well as multi-unit restorations, reflecting a broad range of clinical scenarios. Treatment protocols typically involved combined surgical and regenerative approaches, with GalvoSurge® treatment followed pre-dominantly by non-submerged healing. The mean longest documented follow-up was 19 months, ranging from 8 to 33 months across clinics, with 6 out of 7 clinics reporting the mean follow-up periods of at least 12 months. Participating clinicians were instructed to include all consecutive eligible cases (use of GalvoSurge according to the IFU and at least 6 months of follow-up), irrespective of outcome, to minimize selection bias.
Clinical outcomes demonstrated consistent and statistically significant improvements:
- The PD reduction was observed in 99 implants and ranged from 1 to 10mm. No PD reduction was observed in 5 implants, and none showed an increase in PD.
- The mean ∆PD for 104 treated implants was -3.59 mm.
- The number of implants that did not display BOP increased to 81, while the number of implants with BOP decreased to 23.
These real-world survey data complement published preclinical and clinical evidence and confirm that treatment protocols using GalvoSurge® are associated with consistent, clinically relevant improvements under routine conditions. The observed reductions in probing depth and bleeding on probing, maintained over time and across diverse clinical settings, support the robustness and reproducibility of the approach. Together, the results position GalvoSurge® as a valuable component of contemporary peri-implantitis management strategies.
Key take-aways
- Electrolytic cleaning (GalvoSurge®) is a novel, evidence-supported method for effective implant surface decontamination in peri-implantitis.
- Both clinical studies and real-world data show significant improvements in probing depth, bleeding on probing, and bone regeneration.
- Real-world evidence confirms that this approach is reliable and effective across diverse clinical settings, supporting its integration into routine practice.

